Introduction
Semi-solid extrusion (SSE) 3D printing offers a promising approach for manufacturing personalized medicinal products by enabling precise dose adjustment while avoiding limitations associated with thermolabile active substances and complex pre-processing steps. In contrast to fused deposition modeling, which requires the preparation of drug-loaded filaments, SSE allows direct printing of drug-containing formulations and is therefore well suited for individualized therapies. In this context, real-time detection of process deviations is essential to ensure consistent product quality and patient safety. To address this need, a near-infrared (NIR) probe was integrated directly into the printing nozzle tip, enabling continuous in-line monitoring of the formulation during printing and providing spectral data for process monitoring and quality control.

A New Collaboration to Advance PAT in 3D Printing
To address these challenges, Sentronic has entered a collaboration with Heinrich Heine University Düsseldorf, working closely with PhD student Jonas Hoffmann, whose research focuses on the spectroscopic instrumentation of 3D‑printed, pediatric, individualized formulations produced from semi‑solid matrices. A topic that aligns perfectly with the question of how analytical technologies can be embedded directly into emerging manufacturing platforms. Together, we are exploring how NIR‑based PAT can support the reliable production of personalized medicines in highly compact, decentralized settings.
Integrating PAT into Semi‑Solid Extrusion 3D Printing
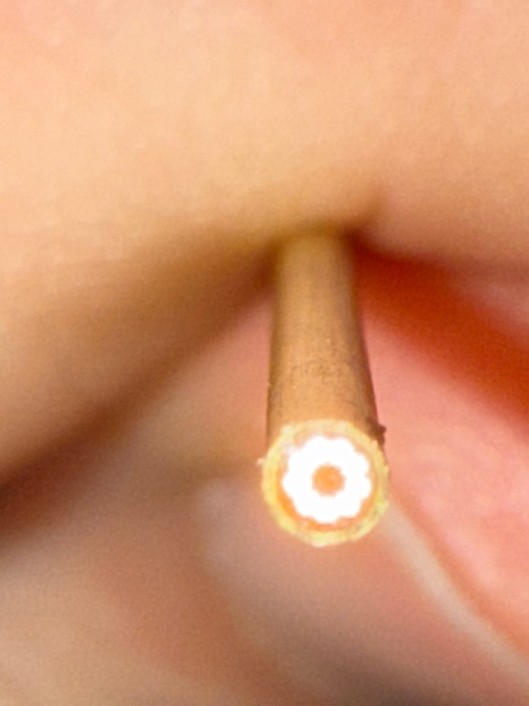
To enable in‑line process monitoring during semi‑solid extrusion (SSE) 3D printing, our Sentronic experts designed a highly compact optical setup that allows a 1.6 mm NIR probe to be positioned directly in the material flow of a standard extrusion nozzle. This required a custom fiber‑optic concept tailored to the extremely limited space available. The resulting configuration features a central collection fiber surrounded by nine illumination fibers, ensuring maximum signal capture despite the tight geometric constraints. Without this dedicated design, PAT integration into such a small nozzle would not have been feasible.
The setup implemented in a 3D‑Bioplotter (EnvisionTEC) illustrates how the printhead is connected to the NIR spectrometer via these custom fiber optics. This arrangement places the measurement point exactly where critical quality attributes emerge, enabling real‑time monitoring of homogeneity, API distribution, and moisture content during the extrusion process.
This work demonstrates how Sentronic’s optical and PAT expertise can bring robust, real‑time analytics into emerging manufacturing technologies. It marks another step toward making additive pharmaceutical production more controlled, transparent, and future‑ready.

First Scientific Presentation and Outlook
This collaborative work was recently presented by Jonas Hoffmann as a scientific poster at the 15th World Meeting on Pharmaceutics, Biopharmaceutics and Pharmaceutical Technology, held 23–26 March 2026 in Prague, Czech Republic. The contribution was met with strong interest from both academic and industrial attendees, highlighting the relevance of integrating PAT into compact, pharmacy‑oriented manufacturing platforms.
Encouraged by this positive response, both partners plan to deepen the scientific work in the coming months and are already preparing a peer‑reviewed publication to share the findings with the broader community.